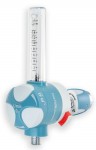
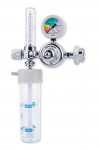
CH, TR and MAK oxygen therapy humidifiers
The bubbling humidifiers for oxygen therapy series CH/200, TR/200, MAK/300 and MAK/500 are devices used to increase the relative humidity in the treatments with medical oxygen, both in hospital and at home.
STRUCTURE
Humidifiers for oxygen series CH/200, TR/200, MAK/300 and MAK/500 are manufactured with both the transparent jar and the cover with bubbler in polycarbonate. All fitments are in brass and they can be sterilized in autoclave at 121 °C for 15 min. The model TR/200 can also be realized in PSU where higher value of sterilization temperature is requested (134 °C - 18 min.).
OPTIONS AND CAPACITIES
The model TR/200, MAK/300 and MAK/500 can be equipped with an optional relief valve to avoid the possible over pressure inside the humidifier. They guarantee, in conjunction with flowmeters EasyFLOW®, Rs, Qmed®, EASYMED® and EasyMED®PLUS, an easy use and great versatility, combined with an extreme economy. They are supplied in different options of capacity (200 ml, 300 ml and 500 ml) and with optional threads in the input connections, this to satisfy the most varied requirements and needs.
Tech Info
|
CH/200 |
TR/200 |
MAK/300 |
MAK/500 |
|
|
Inner volume |
210 ml |
120 ml |
285 ml |
355 ml |
|
Sizes (LxWxH) |
55x67x200 mm |
60x75x190 mm |
70x81x175 mm |
70x81x207 mm |
|
Weight |
0.11 Kg |
0.13 Kg |
0.15 Kg |
0.16 Kg |
|
Max. applicable pressure value |
500 kPa |
|||
|
Max. applicable flow value |
10 L/min. |
|||
|
Inlet connection |
1/4” ISO 3253 F. • 3/8” ISO 3253 F. • M 12x1.25 F. |
|||
|
Outlet connection |
hose connector Ø 6÷9 mm |
|||
|
Relief valve (optional) |
preset at 80 kPa ±10% with a flow equal to 10 L/min. |
|||



Download Area
variable area flowmeters
variable area oxygen flowmeters
variable area oxygen flowmeters
calibrated orifices flowmeters
calibrated orifices oxygen flowmeters
pressure regulators
pressure regulators
pressure regulators for oxygen therapy
Products Oxygen Therapy
-
variable area flowmeters
-
variable area oxygen flowmeters
-
variable area oxygen flowmeters
-
variable area oxygen flowmeters
-
variable area oxygen flowmeters
-
calibrated orifices flowmeters
-
calibrated orifices oxygen flowmeters
-
calibrated orifices oxygen flowmeters
-
flow selector for aerosol therapy
-
pressure regulators
-
pressure regulators
-
pressure regulators for oxygen therapy
-
oxygen therapy humidifiers
-
oxygen therapy humidifiers
-
oxygen therapy humidifiers
-
oxygen therapy humidifiers
-
oxygen therapy humidifiers
-
bubbling humidifiers
-
oxygen therapy single-patient humidifiers
-
-